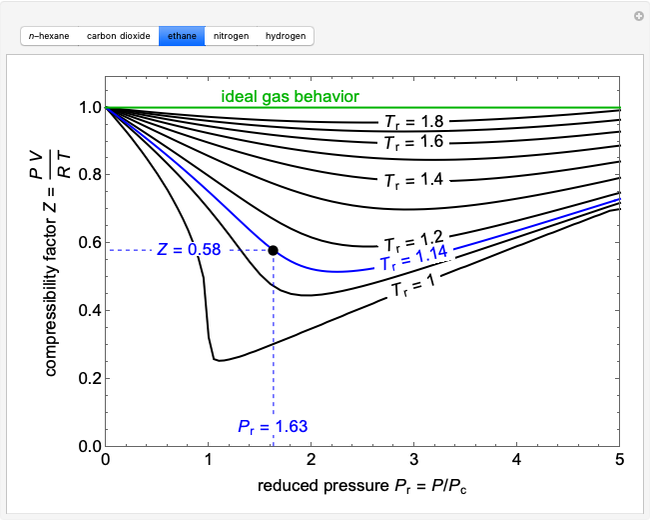
At a high pressure, the compressibility factor (Z) of a real gas is us
By A Mystery Man Writer
Description
At high P. P gt gt (n^(2)a)/(V^(2)) So ‘a’ can be neglected.

Real Gas Behavior The Compression Factor (Z) [Example #2]
The van der Waals equation for one mol of CO(2) gas at low pressure wi

Chemistry Desk: Effect of Pressure

At a high pressure, the compressibility factor (Z) of a real gas is us

The compressibility factor a real gas high pressure is:-1 - frac{Pb} {RT}1 + frac {RT} {Pb}11 + frac {Pb} {RT}

Compressibility factor (z): real gases deviate from ideal behav-Turito

Energies, Free Full-Text

Determine Compressibility of Gases
How does real gas occupies more volume than an ideal gas at high pressure? - Quora

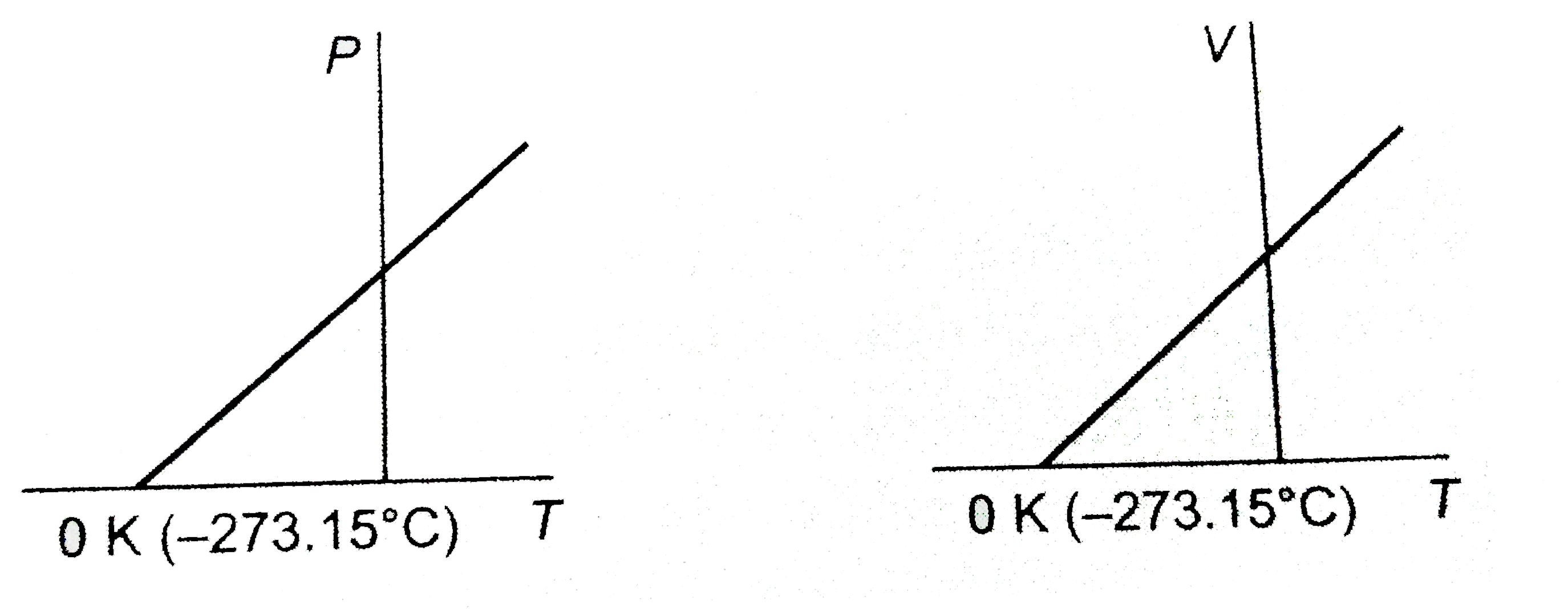
Objectives_template

Explain how the compression factor varies with pressure and

Express the average kinetic energy per mole of a monoatomic gas of mol

At a high pressure, the compressibility factor (Z) of a real gas is usual..
from
per adult (price varies by group size)