2.t 300 K, 36 g of glucose present per litre in itssolution has an
By A Mystery Man Writer
Description
2.t 300 K, 36 g of glucose present per litre in itssolution has an osmotic pressure of 4.98 bar. If theosmotic pressure of solution is 1.52 bar at thesame temperature, what would be itsconcentration?(1) 11 gl 1(3) 36 gl 1(2) 22 gL 1(4) 42 gL 1
2-t 300 K- 36 g of glucose present per litre in itssolution has an osmotic pressure of 4-98 bar- If theosmotic pressure of solution is 1-52 bar at thesame temperature- what would be itsconcentration-1- 11 gl-1-3- 36 gl-1-2- 22 gL-1-4- 42 gL-1
2-t 300 K- 36 g of glucose present per litre in itssolution has an osmotic pressure of 4-98 bar- If theosmotic pressure of solution is 1-52 bar at thesame temperature- what would be itsconcentration-1- 11 gl-1-3- 36 gl-1-2- 22 gL-1-4- 42 gL-1

at 300 Kelvin 30 gram of C6H12O6 present in a litre of its solution has an osmotic pressure of 4.98 bar if

Osmotic pressure of a solution is 0.0821 atm temperature of 300 K . The concentration of solution in moles/litre will be

/wp-content/uploads/2020/09/NCER
Class 12 Chemistry Chapter 2 NCERT Solutions
NCERT Solution for Class 12 Chemistry Chapter 2 - Swastik Classes
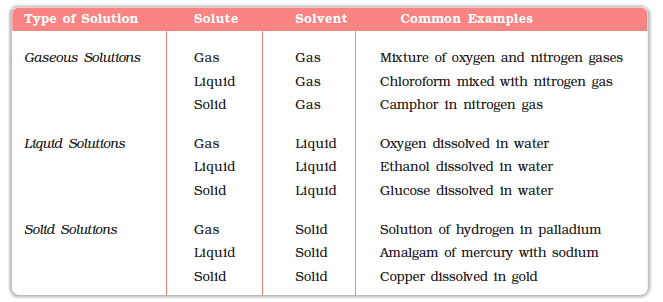
NCERT Ebook for Solutions - Solutions - Chapter 2 - NCERT Chemistry - XII
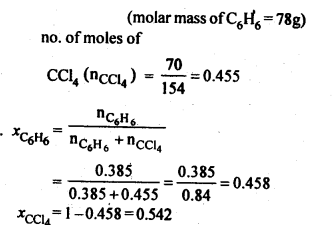
NCERT Solutions For Class 12 Chemistry Chapter 2 Solutions

⏩SOLVED:At 300 K, 36 g of glucose present per litre in its…

SOLUTION: class 12 chemistry quick revision notes - Studypool

At `300 K`, `36 g` of glucose present per litre in its solution has an osmotic pressure of `4.98
from
per adult (price varies by group size)







