Sacituzumab Earns Regular FDA Approval for TNBC - NCI
By A Mystery Man Writer
Description
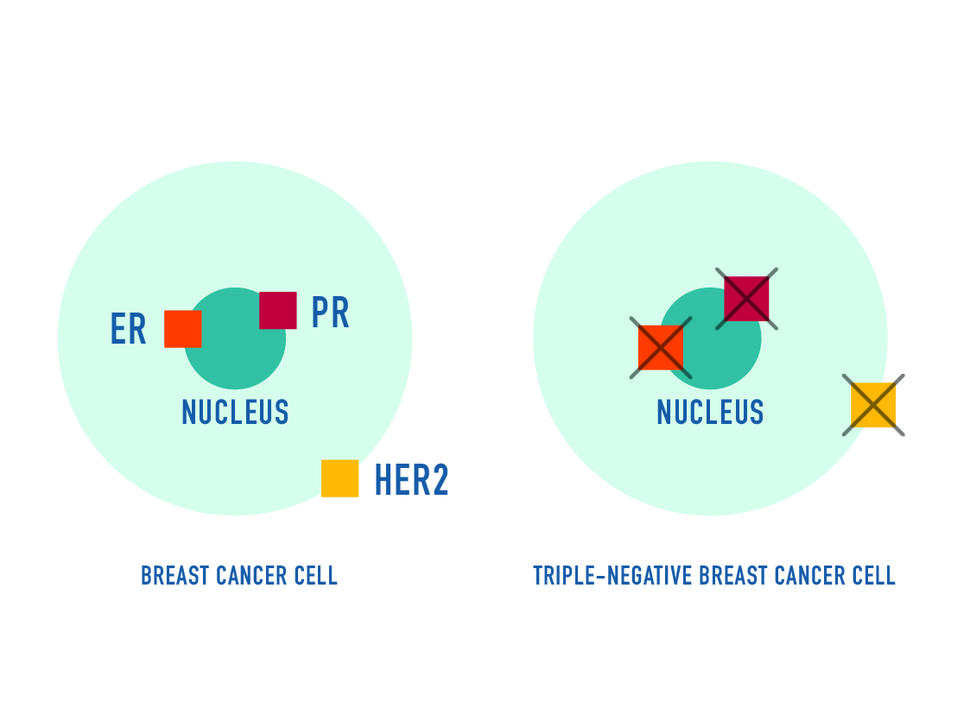
Sacituzumab govitecan (Trodelvy) now has regular FDA approval for people with locally advanced or metastatic triple-negative breast cancer (TNBC), including those with brain metastases. The update follows last year’s accelerated approval of the drug for people with TNBC.

Therapeutic efficacy of IMMU-132 with different DARs. NCI-N87

PDF) Exploiting Therapeutic Vulnerabilities in Triple-Negative
Sacituzumab Earns Regular FDA Approval for TNBC - NCI

FDA Approves Sacituzumab Govitecan for Triple-Negative Breast

Targeting Triple-negative Breast Cancer

PDF) Metastatic Triple-Negative Breast Cancer
View of Sacituzumab Govitecan (Trodelvy) Canadian Journal of Health Technologies

FDA grants accelerated approval to sacituzumab govitecan-hziy for

Pharmaceutics, Free Full-Text

Life Expectancy – Frantic Shanti
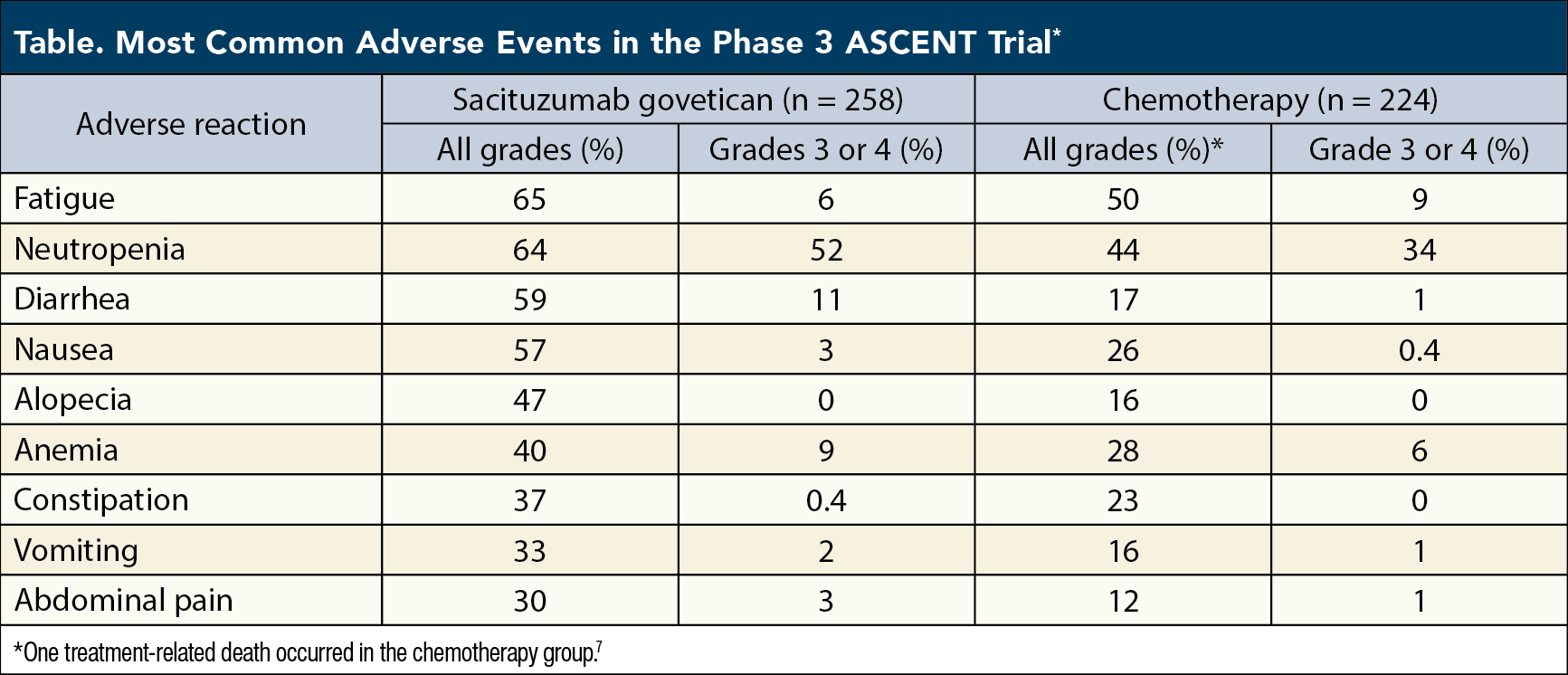
Sacituzumab Govitecan Moves to Second-Line Therapy for Metastatic

Sacituzumab Earns Regular FDA Approval For TNBC NCI

10-K
from
per adult (price varies by group size)







